Diagnosis of neurodegenerative diseases through eye movements
A new robotic system developed by UPM researchers and AURA Innovative Robotics Company can help diagnose neurodegenerative diseases, such as dementia and Parkinson, through the analysis of eye movements.
OSCANN Desk is a non-invasive technology developed by researchers from Universidad Politécnica de Madrid (UPM) and the company AURA Innotive Robotics, led by Cecilia García Cena that with a simple and fast test can provide data about brain functioning through the measurement of eye movements.
This new system is in the phase of clinical trial authorized by the Spanish Agency of Medicines and Medical Devices in six Spanish hospitals and, thanks to techniques of imaging processing and machine learning, its results will allow doctors to early diagnose neurodegenerative diseases and carry out customized treatments.
The diagnosis process of a neurodegenerative disease takes time since symptoms are complex to assess in the early stages of the disease. Besides, there are symptoms that are common to other neurodegenerative diseases such as tremors. High rates of diagnostic uncertainty make objective tests necessary to achieve an accurate medicine in which each patient receives information, prognosis and appropriate treatment.
Credit: AURA Innovative Robotics
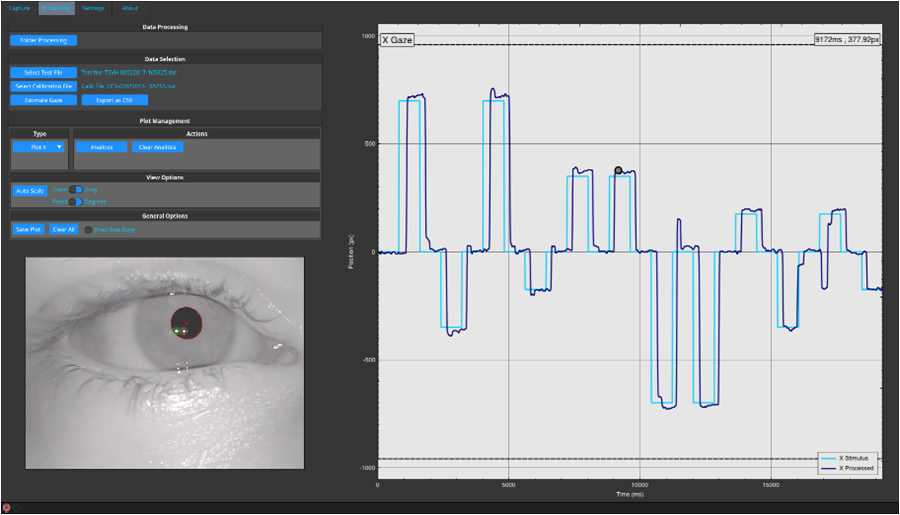
The physiological process in medicine explains the eye movements. To accurately measure these movements would provide real-time information about how the brain is working at that moment. From this premise and in order to achieve an early diagnosis of neurodegenerative diseases, researchers from Centre for Automation and Robotics (CAR) CSIC-UPM and AURA Innovative Robotics Startup Company have developed OSCANN desk, an assistant medical device that through techniques of image processing and machine learning is able to accurately assess the eye movements.
Thanks to this new tool, doctors will have objective data of the brain functioning that, along with other clinical data, will use to carry out an accurate early diagnosis of the disease.
The test is conducted in a health care center with no need for a second doctor's appointment. The personnel select a set of calibration and tests of each patient. The patient sits comfortably in a chair and the device is adapted to his anatomy to precisely measure the eye movement. The patient has to look at the stimulus that appears on a monitor, each test lasts about a minute.
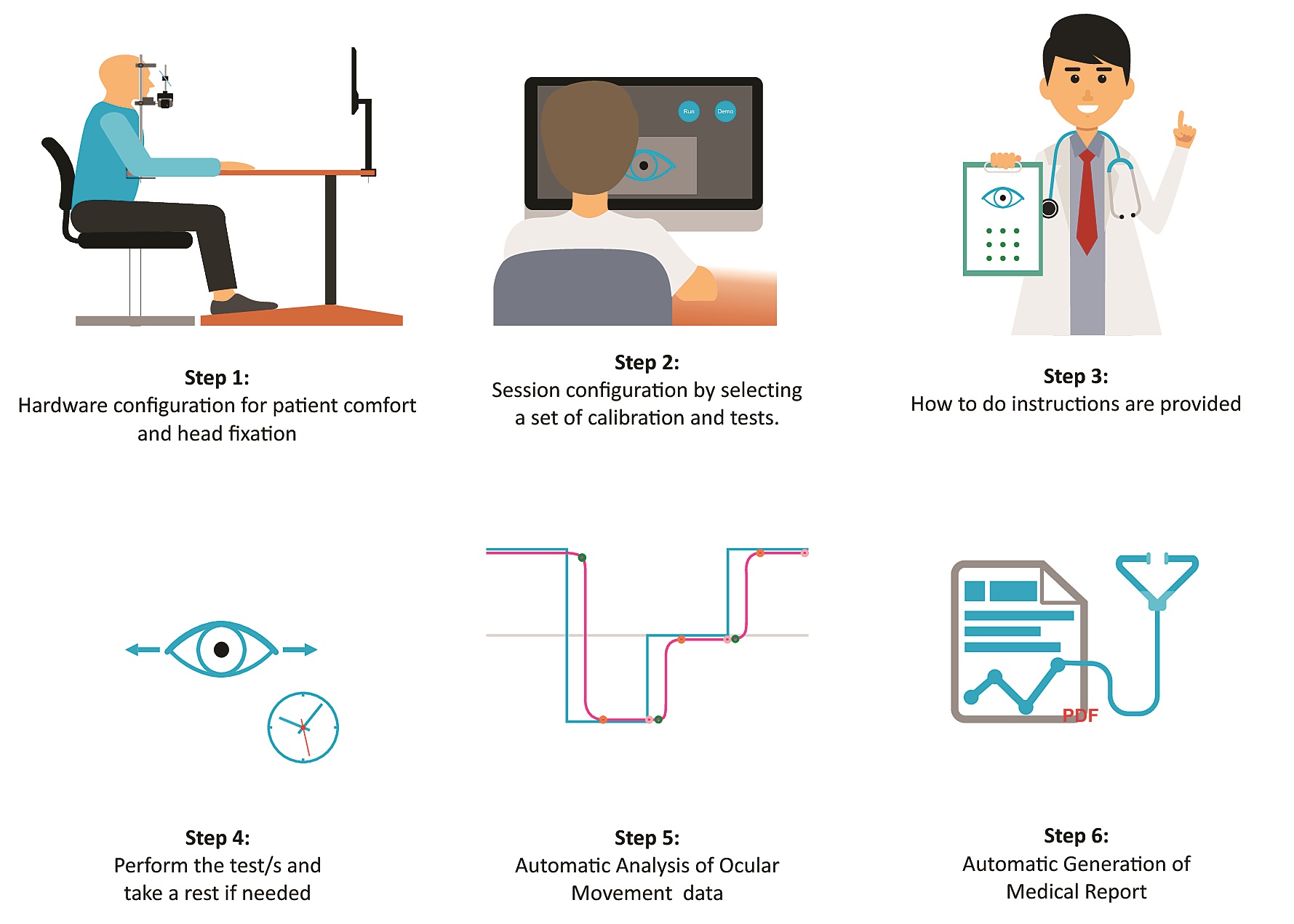
The process using OSCANN desk. Credit: UPM
The clinical tests allowed researchers to develop models of pathologies and, by applying machine learning techniques, similarities and differences are searched among over 500 variables of eye movement. Likewise, the progress of certain symptoms can be objectively measured. This will help doctors to make a diagnosis and customize the treatment.
Today, the tests are applied to Alzheimer's, Parkinson's, mild cognitive impairment, diverse dementias, multiple sclerosis, etc. Besides, researchers are collaborating in other clinical research such as autistic spectrum disorders, epilepsy, diabetes, alcoholism, migraines, depression and bipolar disorder. This tool is being used in six hospitals that are national reference centers in pathologies: Hospital Universitario 12 de Octubre, Hospital Sant Pau, Hospital Clinic de Valencia, Hospital Clinic de Barcelona, Hospital Marqués de Valdecilla and Hospital Valle de Hebrón. Besides, the Centro de Investigaciones Príncipe Felipe is collaborating in the project.
In a near future, OSCANN Desk will be working in HM Hospitals, specifically in the Memory Disorders Unit of HM CINAC located at the Hospital Universitario HM Madrid.
According to the main researcher of this work: “the DNA used to develop this technology has a vital gene: the accessibility. The cost should not be an impediment to use the OSCANN desk technology by those who need it”.
Hernandez, Erik; Hernandez, Santiago; Molina, David; Acebron, Rafael; Garcia Cena, Cecilia E. OSCANN: Technical Characterization of a Novel Gaze Tracking Analyzer SENSORS, 18 (2):10.3390/s18020522 FEB 2018


